I'm "reconditioning" a Nickel Cadmium 2.5Ah battery pack out of a piece of test equipment I'm repairing for someone. The pack is made up of 10 cells and was found at 0V when I first measured it.
Initially I put 100mA through them and checked for major shorts. They seemed OK so I gradually increased the current to half an amp and checked the temperature (finger thermometer) of the cells as the voltage increased. Only a slightly perceptible increase in temp above ambient on a few of them. Once the voltage reached 13V I disconnected the current supply and the voltage held.
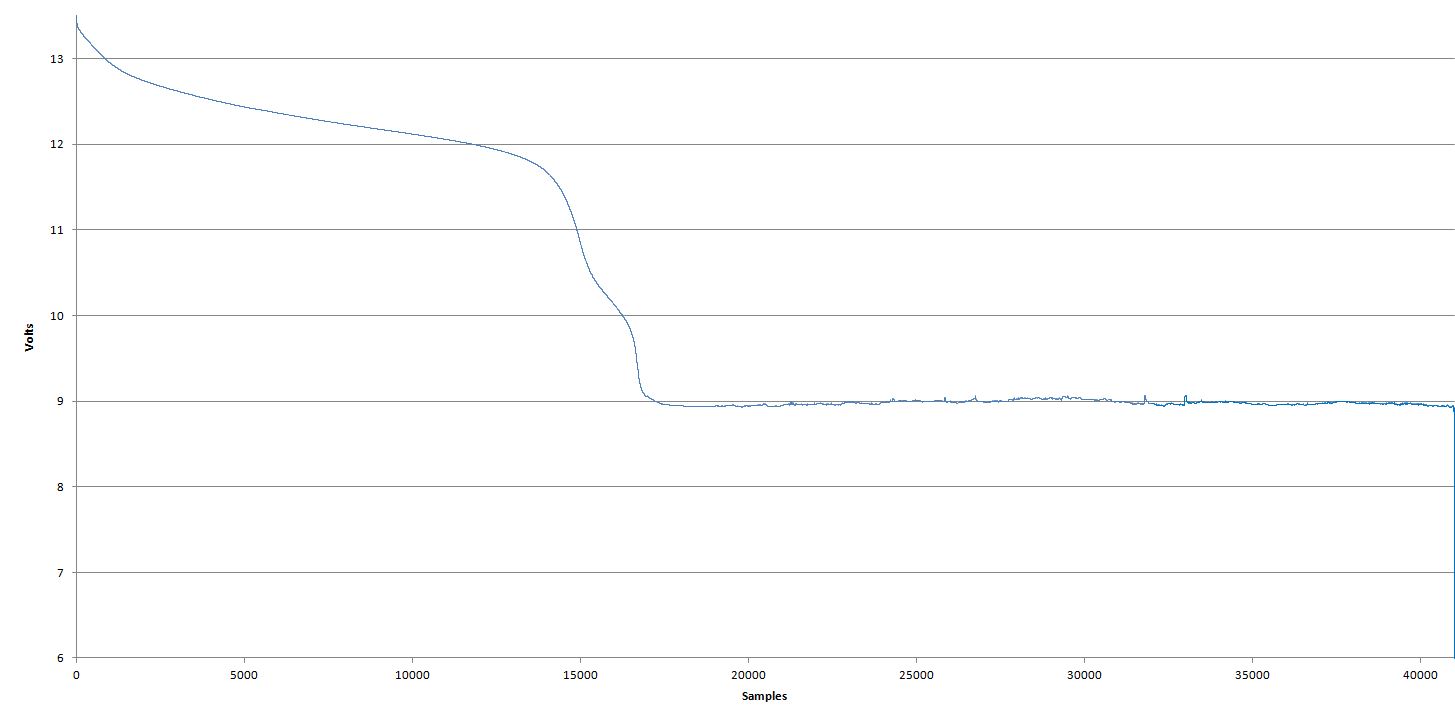
I then connected the pack up to my Maynuo M9812 dc load, fired up its software set the safe voltage to 6V (0.6V per cell) and drew 1A out of them. The pack quickly ( a few minutes) dropped down to about 8.8V where it sat for the majority of the discharge. Whilst sitting around this voltage and drawing 1A I started seeing the terminal voltage
increase. Not much mind you, around 300 to 500 mV so from 8.7 to 9.3V all whilst drawing 1A
I haven't played much with NiCd's before and certainly wasn't expecting to see a battery's voltage increase whilst discharging it. Anybody know what's going on here? I don't know whether this something to do with the chemistry or a mix of damaged/unbalanced cells coming to some form of equilibrium